 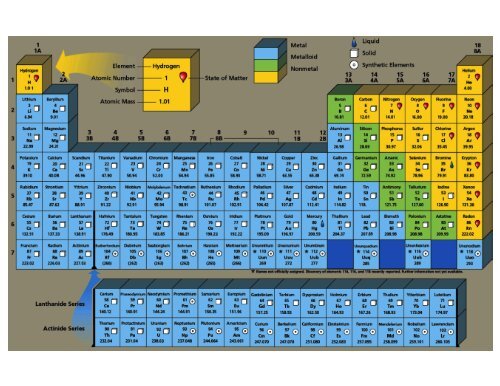 
Note that some metal atoms, specifically the transition elements, commonly form ions with variable charges. The charge that a representative (main group element) acquires when it becomes an ion is related to the location on the periodic table. Thus, the periodic table becomes a tool for remembering the charges on many ions. 1: Common ions formed by specific elements on the periodic table. So, the compound is silver(I) chloride, but it is commonly known as silver. 
\)).īecause it is impossible to measure the sizes of both metallic and nonmetallic elements using any one method, chemists have developed a self-consistent way of calculating atomic radii using the quantum mechanical functions. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge. Refer to the periodic table above, Ag forms 1+ charge and Cl forms 1- charge. The ionic charge trend in the periodic table is that generally elements on the left form cations and those on the right form anions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
